Careful investigations have shown that not all electrons within an atom have the same average position or energy. The electrostatic attraction between them keeps electrons ‘bound’ to the nucleus so they stay within a certain distance of it. Principal Energy LevelsĪn atom consists of a positively charged nucleus and negatively charged electrons. These Lewis symbols and Lewis structures help visualize the valence electrons of atoms and molecules, whether they exist as lone pairs or within bonds. Lewis structures (also known as Lewis dot structures or electron dot structures) are diagrams that represent the valence electrons of atoms within a molecule. Lewis symbols (also known as Lewis dot diagrams or electron dot diagrams) are diagrams that represent the valence electrons of an atom. Lewis symbols: Symbols of the elements with their number of valence electrons represented as dots.valence electrons: The electrons of atoms that participate in the formation of chemical bonds.valence level: The outermost principal energy level, which is the level furthest away from the nucleus that still contains electrons. 
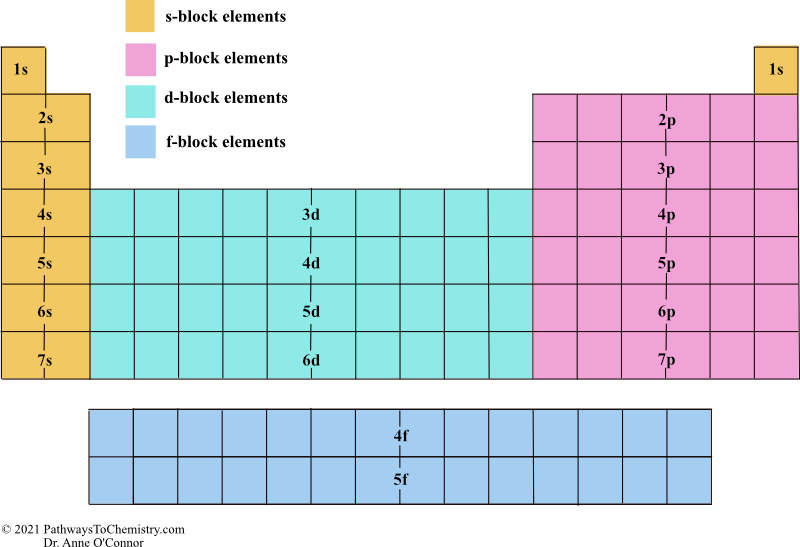
Each level is associated with a particular energy value that electrons within it have. principal energy levels: The different levels where electrons can be found and that occur at specific distances from the atom’s nucleus.Lewis symbols do not visualize the electrons in the inner principal energy levels.Lewis symbols are diagrams that show the number of valence electrons of a particular element with dots that represent lone pairs.The outermost principal energy level that contains electrons is called the valence level and contains valence electrons.Electrons exist outside of an atom ‘s nucleus and are found in principal energy levels that contain only up to a specific number of electrons.For more information on how electron configurations and the periodic table are linked, visit the Connecting Electrons to the Periodic Table module. The periodic table is an incredibly helpful tool in writing electron configurations. Using the periodic table to determine the electron configurations of atoms is key, but also keep in mind that there are certain rules to follow when assigning electrons to different orbitals. The s-block is the region of the alkali metals including helium (Groups 1 & 2), the d-block are the transition metals (Groups 3 to 12), the p-block are the main group elements from Groups 13 to 18, and the f-block are the lanthanides and actinides series. Each orbital can be represented by specific blocks on the periodic table. The 1s orbital and 2s orbital both have the characteristics of an s orbital (radial nodes, spherical volume probabilities, can only hold two electrons, etc.) but, as they are found in different energy levels, they occupy different spaces around the nucleus. 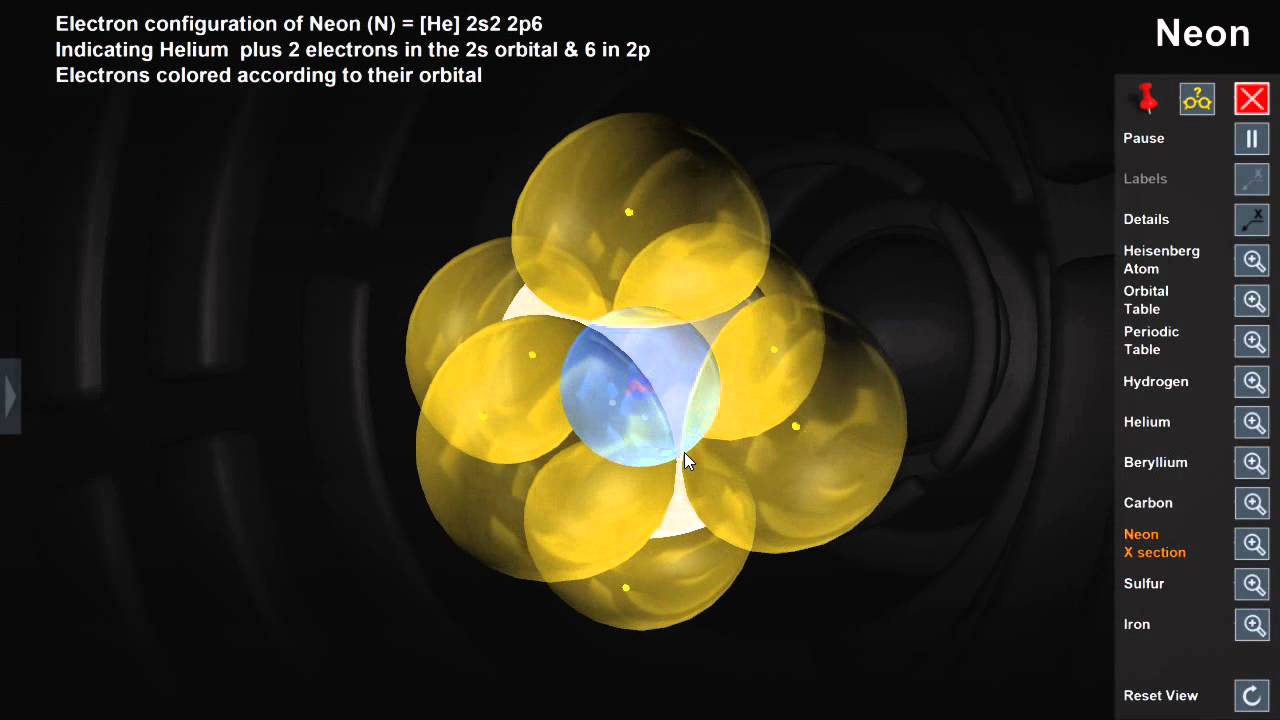
Orbitals on different energy levels are similar to each other, but they occupy different areas in space. The energy level is determined by the period and the number of electrons is given by the atomic number of the element. The p, d, and f orbitals have different sublevels, thus can hold more electrons.Īs stated, the electron configuration of each element is unique to its position on the periodic table. The four different types of orbitals (s,p,d, and f) have different shapes, and one orbital can hold a maximum of two electrons. Electrons exhibit a negative charge and are found around the nucleus of the atom in electron orbitals, defined as the volume of space in which the electron can be found within 95% probability. Every element on the Periodic Table consists of atoms, which are composed of protons, neutrons, and electrons. \)īefore assigning the electrons of an atom into orbitals, one must become familiar with the basic concepts of electron configurations.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
